 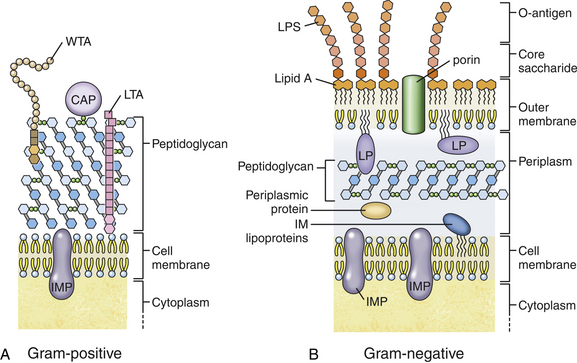  
Furthermore, the byproducts of gram-negative bacteria, such as lipopolysaccharides (LPS), capsular proteins, fimbrillins and flagellins, can also penetrate the brain and affect neuroinflammation, and Aβ and tau pathology. In particular, several gram-negative bacteria, such as Proteobacteria and Chlamydophila pneumonia, have been reported to provoke strong systemic inflammation and contribute to AD pathogenesis. In addition, it has been reported that the gram-negative bacteria can cross the blood–brain barrier (BBB) and contribute to neuroinflammation, Aβ accumulation, and tau hyperphosphorylation within the brain. The brains of AD patients contain 5–10 times more bacteria than healthy brains differences in the distribution and composition of the bacteria have also been studied. Surprisingly, systemic inflammatory responses due to bacterial infection may also continue, causing neuronal cell death and Aβ/tau accumulation, contributing to the development and progression of AD. The occurrence of microbiome dysbiosis or infection of several species of toxic bacteria may contribute to AD pathogenesis by triggering strong inflammatory responses or participating in Aβ production. 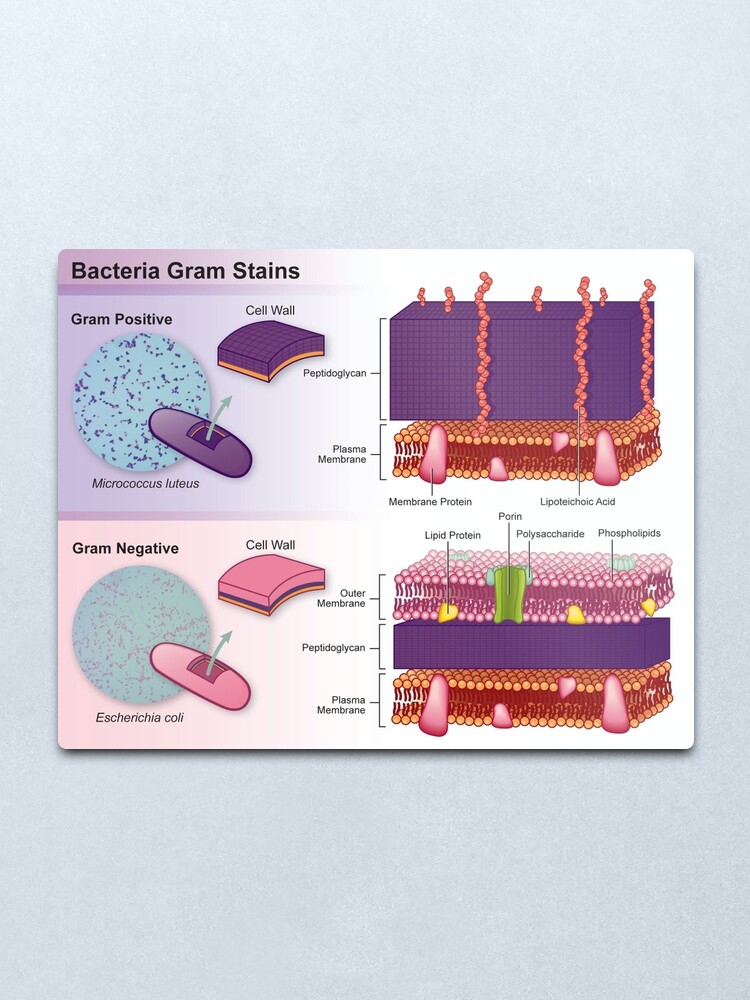
Various sources of infection, such as fungi, viruses, and bacteria, are reportedly associated with AD. Namely, extensive changes in the microbiome occur in AD, and studies analyzing the intestinal microbiome in AD patients and animal models have provided interesting insights. Many studies regarding the relationships between microbes and AD have indicated microorganisms as one of the new therapeutic targets for AD. Therefore, changes in therapeutic targets are required, and upstream pathogenic contributors that affect Aβ and tau pathology are receiving increasing interest. Medications targeting Aβ and tau have been developed to treat AD however, they have not been effective in clinical trials. Previous studies have demonstrated that amyloid-β (Aβ) and tau are pathological hallmarks and primary causes of AD. Furthermore, we discuss the possibility of using gram-negative bacteria and gram-negative bacterial molecules as novel therapeutic targets and new pathological characteristics for AD.Īlzheimer's disease (AD), one of the main causes of dementia, is a neurodegenerative disease causing cognitive decline and impairment of memory, language, and attention. Here, we summarize the roles and pathomechanisms of gram-negative bacteria and LPS in AD. Despite multiple evidence showing that the gram-negative bacteria and their LPS play a crucial role in AD pathogenesis, the pathogenic mechanisms of gram-negative bacteria and their LPS have not been clarified. Moreover, therapeutic approaches targeting gram-negative bacteria or gram-negative bacterial molecules have significantly alleviated AD-related pathology and cognitive dysfunction. Moreover, gram-negative bacteria and their LPS have been shown to affect a variety of AD-related pathologies, such as Aβ homeostasis, tau pathology, neuroinflammation, and neurodegeneration. Surprisingly, an altered distribution of gram-negative bacteria and their LPS has been reported in AD patients. The gram-negative bacteria and lipopolysaccharides (LPS) are attractive new targets for AD treatment. Failures in the development of therapeutics targeting amyloid-β (Aβ) and tau, principal proteins inducing pathology in AD, suggest a paradigm shift towards the development of new therapeutic targets. Alzheimer’s disease (AD) is the most serious age-related neurodegenerative disease and causes destructive and irreversible cognitive decline. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
